Single-layer Cartridge Filter (PES 0.45μm)
Product Introduction
Alioth Alipore® CHF PES prefilters can provide effective bioburden control and particle contaminants removal with good chemical compatibility and low level extractable. Alipore® CHF filters aresuitable for bioburden control and prefiltration application of various kind of fluid,provide effective protection to downstream sterilizing-grade filter and other processsystem in different steps of bioprocess. Meet the common needs of the pharmaceutical industry for liquid pre-filtration.
Key Features and Benefits
- High flow rate, high capacity
- Valicated bioburden reduction
- Stable performance, Withstand multiple
- cycles of sterilization
- Broad chemical compatibility
Typical Applications
- Pre-fltration for buffers
- Cell culture media
- Pre-filtration for column protection
- Pre-filtration before terminal sterilization, etc
- Alioth Filter Series
- Differential
Typical Flow Characteristics
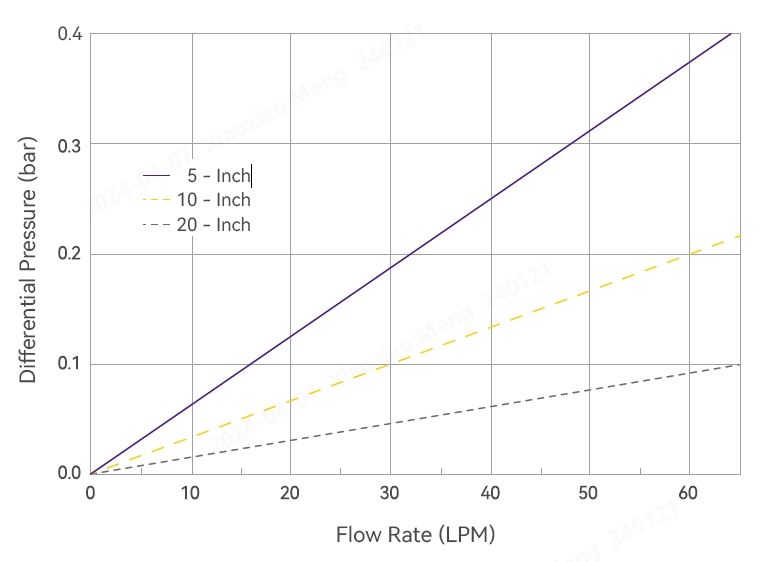
Product Specifications
| 5 inch | 10 inch | 20 inch | 30 inch | |
|
Dimensions Diameter |
69 mm |
69 mm |
69 mm |
69 mm |
| Filtration Area | 0.32 m2 | 0.65 m2 | 1.3 m2 | 1.95 m2 |
| Materials of Construction |
Filter membrane: Hydrophilic polyethersulfone (PES) Supporting Layer: Polypropylene (PP) Liner/Cartridge body: Polypropylene (PP) |
|||
| Pore Size | 0.45 μm | |||
| Maximum Differential Pressure |
Forward: 5.0 bar ( 72.5 psi ) @ 25℃ / 3.0 bar ( 43.5 psi ) @ 80℃ / Reverse: 2.0 bar ( 29.0 psi ) @ 25℃ | |||
| Sterilization Parameters | 135℃, 30 minutes, 10 cycles of high-pressure sterilization or 10 cycles of sterilization in place | |||
| Biological Safety | All the construction components of this cartridge comply with the requirements for biological safety of Class Vl plastics in current USP < 88 > | |||
| Indirect Food Additive | The primary construction components of this cartridge comply with the reguirements for food contact materials as stipulated in EU 1935 / 2004 / EC and FDA 21 CFR 177-182 | |||
| Cleanliness | Cleanliness meets the requirements for nonfiber releasing filter as specified in FDA 21 CFR 211.72 and 210.3 (b) (5) (6), and the level of insoluble particles in rinsing liquid meets the requirements of USP < 788 > | |||
| Endotoxin | The endotoxin level of cartridge rinsing liquid is < 0.25 EU/mL as indicated by gel method, which complies with the requirements of USP < 85 > | |||
| TOC / Conductivity | TOC < 0.5 mg/L, Conductivity < 1.3 μS/cm | |||
| Manufacturing Environment | Manufactured in conformance with cGMP | |||
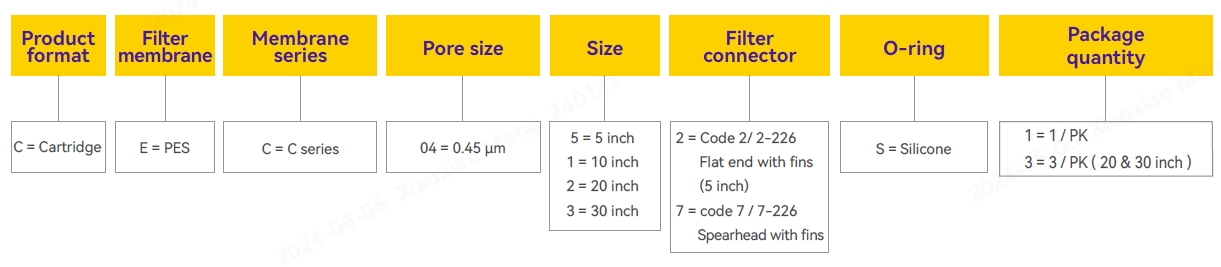
Ordering Information
For further information, please contact us and Alioth technical team will provide a comprehensive service for your filtration process.


